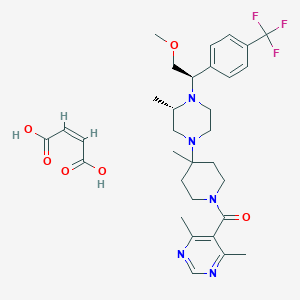
-
Vicriviroc Maleate
- names:
Vicriviroc Maleate
- CAS号:
599179-03-0
MDL Number: Not available - MF(分子式): C32H42F3N5O6 MW(分子量): 649.701
- EINECS:Not available Reaxys Number:Not available
- Pubchem ID:6451165 Brand:BIOFOUNT
| 货品编码 | 规格 | 纯度 | 价格 (¥) | 现价(¥) | 特价(¥) | 库存描述 | 数量 | 总计 (¥) |
|---|---|---|---|---|---|---|---|---|
| HCC352719-2.5mg | 2.5mg | 97% | ¥ 1995.00 | ¥ 1995.00 | 4-7周 | ¥ 0.00 |
| 中文别名 | Vicriviroc Maleate(599179-03-0);sch 417690;sch-417690;Sch417690;1-((4,6-二甲基-5-嘧啶基)羰基)-4-(4-(2-甲氧基-4-(三氟甲基)苯基)乙基-3-甲基-1-哌嗪基)-4-甲基哌啶; |
| 英文别名 | Vicriviroc Maleate(599179-03-0);Sch 417690;Sch-417690;Sch417690;vicriviroc;1-((4,6-dimethyl-5-pyrimidinyl)carbonyl)-4-(4-(2-methoxy-4-(trifluoromethyl)phenyl)ethyl-3-methyl-1-piperazinyl)-4-methylpiperidine; |
| CAS号 | 599179-03-0 |
| Inchi | InChI=1S/C28H38F3N5O2.C4H4O4/c1-19-16-35(14-15-36(19)24(17-38-5)22-6-8-23(9-7-22)28(29,30)31)27(4)10-12-34(13-11-27)26(37)25-20(2)32-18-33-21(25)3;5-3(6)1-2-4(7)8/h6-9,18-19,24H,10-17H2,1-5H3;1-2H,(H,5,6)(H,7,8)/b;2-1-/t19-,24-;/m0./s1 |
| InchiKey | GXINKQQWHLIBJA-UCIBKFKQSA-N |
| 分子式 Formula | C32H42F3N5O6 |
| 分子量 Molecular Weight | 649.701 |
| 溶解度Solubility | 生物体外In Vitro:DMSO溶解度50 mg/mL(76.96 mM;Need ultrasonic)H2O : 25 mg/mL(38.48 mM;Need ultrasonic and warming) |
| 性状 | 白色至灰白色固体粉末 |
| 储藏条件 Storage conditions | -20°C Refrigerator |
Vicriviroc Maleate(599179-03-0,sch 417690)实验注意事项:
1.实验前需戴好防护眼镜,穿戴防护服和口罩,佩戴手套,避免与皮肤接触。
2.实验过程中如遇到有毒或者刺激性物质及有害物质产生,必要时实验操作需要手套箱内完成以免对实验人员造成伤害
3.实验后产生的废弃物需分类存储,并交于专业生物废气物处理公司处理,以免造成环境污染Experimental considerations:
1. Wear protective glasses, protective clothing and masks, gloves, and avoid contact with the skin during the experiment.
2. The waste generated after the experiment needs to be stored separately, and handed over to a professional biological waste gas treatment company to avoid environmental pollution.
Tags:Vicriviroc Maleate试剂,Vicriviroc Maleate杂质,Vicriviroc Maleate中间体,Vicriviroc Maleate结构式,Vicriviroc Maleate溶解度,Vicriviroc Maleate密度,Vicriviroc Maleate旋光度,Vicriviroc Maleate闪点,Vicriviroc Maleate购买,Vicriviroc Maleate熔点,
| 产品说明 | Vicriviroc Maleate(599179-03-0,sch 417690)可以作为药物杂质对照品以及生物医药类试剂。 |
| Introduction | Vicriviroc Maleate(599179-03-0,sch 417690)can be used as a reference substance for drug impurities and reagents,only for research. |
| Application1 | sch 417690是Vicriviroc的马来酸盐形式,Vicriviroc是一种基于哌嗪的CCR5受体拮抗剂,具有抗人免疫缺陷病毒的活性。Vicriviroc旨在与CCR5结合并抑制HIV进入CD4细胞。 |
| Application2 | |
| Application3 |
Vicriviroc Maleate(599179-03-0,sch 417690)药理学:
Vicriviroc Maleate(Sch-417690)是一种CCR5拮抗剂,在临床开发中用于治疗HIV-1的IC50为0.91 nM。; IC50值:0.91 nM;目标:CCR5;进行了两项I期研究以评估Vicriviroc的安全性。马来酸Vicriviroc目前正处于晚期临床开发中,这是用于HIV-1感染者的利托那韦增强蛋白酶抑制剂治疗方案的一部分。在临床研究中,Vicriviroc已显示出有效而持久的病毒学抑制作用,免疫活性和普遍良好的耐受性。
Vicriviroc Maleate是一种有效的,选择性,可口服,可透过血脑屏障的CCR5拮抗剂,Ki约为2.5 nM,同时抑制跨血单核细胞中的HIV-1的活性,IC90 值分别为3.3 nM(JrFL),2.8 nM(ADA-M),1.8 nM(301657),4.9 nM(JV1083)和10 nM(RU 570)。
Vicriviroc Maleate是CC趋化因子CCR5受体的口服,有效且选择性的变构拮抗剂。Vicriviroc抑制HIV-1与CCR5受体的相互作用,从而防止HIV-1病毒进入细胞。
| 警示图 | |
| 危险性 | warning |
| 危险性警示 | Not available |
| 安全声明 | H303+H313+H333 |
| 安全防护 | P264+P280+P305+P351+P338+P337+P313 |
| 备注 | 实验过程中防止吸入、食入,做好安全防护 |
| 象形图 |  |
| 信号 | Warning |
| GHS危险说明 | Aggregated GHS information provided by 35 companies from 1 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. |
| H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral] | |
| H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation] | |
| H373 (100%): Causes damage to organs through prolonged or repeated exposure [Warning Specific target organ toxicity, repeated exposure] | |
| Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. | |
| 防范说明代码 | P260, P264, P270, P280, P301+P312, P305+P351+P338, P314, P330, P337+P313, and P501 |
| (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Tagat JR, et al. Piperazine-based CCR5 antagonists as HIV-1 inhibitors. IV. Discovery of 1-[(4,6-dimethyl-5-pyrimidinyl)carbonyl]- 4-[4-[2-methoxy-1(R)-4-(trifluoromethyl)phenyl]ethyl-3(S)-methyl-1-pi |
| Strizki JM, et al. Discovery and characterization of vicriviroc (SCH 417690), a CCR5 antagonist with potent activity against human immunodeficiency virus type 1. Antimicrob Agents Chemother. 2005 Dec; |
| Synthesis of (3) H, (2) H4 and (14) C-SCH 417690 (Vicriviroc) PMID 26991320; Journal of labelled compounds & radiopharmaceuticals 2016 May; 59(5):190-6 Name matches: ccr5 sch 417690 |
| Discovery and characterization of vicriviroc (SCH 417690), a CCR5 antagonist with potent activity against human immunodeficiency virus type 1 PMID 16304152; Antimicrobial agents and chemotherapy 2005 |
| Human immunodeficiency virus (HIV) entry inhibitors (CCR5 specific blockers) in development: are they the next novel therapies? PMID 16306033; HIV clinical trials 2005 Sep; 6(5):272-7 Name matches: cc |
Vicriviroc Maleate(599179-03-0,sch 417690)参考文献:
1.Gateways to clinical trials.
Tomillero A, Moral MA. Methods Find Exp Clin Pharmacol. 2009 Sep;31(7):463-93.
Gateways to Clinical Trials is a guide to the most recent clinical trials in current literature and congresses. The data in the following tables has been retrieved from the Clinical Trials Knowledge Area of Prous Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issue focuses on the following selection of drugs: AAV1/SERCA2a, Abacavir sulfate/lamivudine, Adalimumab, Aliskiren fumarate, Ambrisentan, Aripiprazole, AT-7519, Atazanavir sulfate, Atomoxetine hydrochloride, Azacitidine, Azelnidipine; Besifloxacin hydrochloride, Bevacizumab, Bioabsorbable everolimus-eluting coronary stent, Bortezomib, Bosentan, Budesonide/formoterol fumarate; CAIV-T, Carisbamate, Casopitant mesylate, Certolizumab pegol, Cetuximab, Ciclesonide, Ciprofloxacin/dexamethasone, CTCE-9908; Dalcetrapib, Darunavir, Deferasirox, Desloratadine, Disitertide, Drotrecogin alfa (activated), DTA-H19, Duloxetine hydrochloride, Dutasteride; Ecogramostim, Efalizumab, Emtricitabine, Eribulin mesilate, Escitalopram oxalate, Eszopiclone, EUR-1008, Everolimus-eluting coronary stent, Exenatide; Fampridine, Fluticasone furoate, Formoterol fumarate/fluticasone propionate, Fosamprenavir calcium, Fulvestrant; Gabapentin enacarbil, GS-7904L; HPV-6/11/16/18, Human Secretin, Hydralazine hydrochloride/isosorbide dinitrate; Imatinib mesylate, Imexon, Inalimarev/Falimarev, Indacaterol, Indacaterol maleate, Inhalable human insulin, Insulin detemir, Insulin glargine, Ixabepilone; L-Alanosine, Lapatinib ditosylate, Lenalidomide, Levocetirizine dihydrochloride, Liraglutide, Lisdexamfetamine mesilate, Lopinavir, Loratadine/montelukast sodium, Lutropin alfa; MeNZB, Mepolizumab, Micafungin sodium, Morphine hydrochloride; Nabiximols, Nikkomycin Z; Olmesartan medoxomil, Omalizumab; Paclitaxel-eluting stent, Pegfilgrastim, Peginterferon alfa-2a, Peginterferon alfa-2b, Perifosine, PF-489791, Plitidepsin, Posaconazole, Pregabalin; QAX-576; Raltegravir potassium, Ramelteon, Rasagiline mesilate, Recombinant human relaxin H2, rhGAD65, Rivaroxaban, Rosuvastatin calcium, Rotigotine; Saxagliptin, SCH-530348, Sirolimus-eluting stent, SLIT-amikacin, Sorafenib, Sotrastaurin, SR-16234, Sulforaphane; Tadalafil, Tanespimycin, Tapentadol hydrochloride, Teriparatide, Tesofensine, Tiotropium bromide, Tipifarnib, Tirapazamine, TMC-207, Tocilizumab, Tolvaptan, Tosedostat, Treprostinil sodium; Ustekinumab; Varespladib methyl, Vicriviroc, Vildagliptin, Vildagliptin/metformin hydrochloride, Volociximab, Voriconazole; Ziconotide, Ziprasidone hydrochloride.
2.Gateways to clinical trials.
Tomillero A, Moral MA. Methods Find Exp Clin Pharmacol. 2010 Jun;32(5):331-88. doi: 10.1358/mf.2010.32.5.1520420.
[¹¹C]RAC; (18)F-Fluoromisonidazole; 89-12; 9-[¹?F]Fluoropropyl-(+)-dihydrotetrabenazine; Adalimumab, Adecatumumab, ADMVA, ADXS-11-001, Aflibercept, Agatolimod sodium, AGS-004, Alglucosidase alfa, Aliskiren fumarate, Alvocidib hydrochloride, AMG-108, AMG-853, Apixaban, Aripiprazole, Armodafinil, Atazanavir sulfate, Atomoxetine hydrochloride; Bevacizumab, BioMatrix Flex drug eluting stent, Biphasic insulin aspart, Bortezomib, Bosentan; Caspofungin acetate, Cediranib, Cetuximab, ChimeriVax-Dengue, Choriogonadotropin alfa, Cinacalcet hydrochloride, Cizolirtine citrate, Clofarabine, Cocaine conjugate vaccine, CX-717; Darbepoetin alfa, Dasatinib, Decitabine, Denosumab, Desvenlafaxine succinate, Dexamethasone sodium phosphate, Dienogest, Diphencyprone, Doripenem, DTaP-HepB-IPV, Dutasteride; E-7010, Ecallantide, Ecstasy, Eicosapentaenoic acid/docosahexaenoic acid, Emtricitabine, Enfuvirtide, Erlotinib hydrochloride, Eszopiclone, Etonogestrel/ethinyl estradiol, Etoricoxib, Everolimus, Everolimus-eluting coronary stent EVT-201, Ezetimibe, Ezetimibe/simvastatin; Ferumoxytol, Fesoterodine fumavate, Figitumumab, Filgrastim, Fingolimod hydrochloride, Fluticasone furoate, Fluval P, Fluzone, Fondaparinux sodium, Fulvestrant, Fungichromin; Gamma-hydroxybutyrate sodium, Gefitinib, GHB-01L1, GLY-230, GSK-1349572; Hib-MenCY-TT, Hib-TT, HPV-6/11/16/18, Hydrocodone bitartrate; IC-51, Icatibant acetate, Imatinib mesylate, Immunoglobulin intravenous (human), Indetanib, Influenza A (H1N1) 2009 Monovalent Vaccine, Inhalable human insulin, Insulin glargine, Insulin glulisine, Interferon-beta, Ispinesib mesylate, Ixabepilone; Laromustine, Latanoprost/timolol maleate, L-Citrulline, Lenalidomide, Lexatumumab, Linezolid, Lopinavir/ritonavir, Lutropin alfa; Mapatumumab, MDX-066, MDX-1388, Mepolizumab, Methoxy polyethylene glycol-epoetin-beta, Metreleptin, Micafungin sodium, Mometasone furoate/oxymetazoline hydrochloride, Mx-dnG1, Mycophenolic acid sodium salt; Nabiximols, Natalizumab, Nemonoxacin, Norelgestromin/ethinyl estradiol; Oblimersen sodium, Ocriplasmin, Olmesartan medoxomil, Omacetaxine mepesuccinate; Paclitaxel-eluting stent, Pagoclone, Paliperidone, Panitumumab, Pazopanib hydrochloride, PCV7, Pegaptanib octasodium, Peginterferon alfa-2a, Peginterferon alfa-2b/ ribavirin, Pegvisomant, Pemetrexed disodium, Perifosine, Pimecrolimus, Pitavastatin calcium, Plerixafor hydrochloride, Plitidepsin, Posaconazole, Pregabalin, Progesterone capriate; Raltegravir potassium, Ramucirumab, Ranelic acid distrontium salt, Rasburicase, Recombinant Bet V1, Recombinant human insulin, rhFSH, Rolofylline, Romidepsin, Romiplostim, Rosuvastatin calcium; Sapacitabine, Sevelamer carbonate, Sinecatechins, Sirolimus-eluting stent, Sitagliptin phosphate monohydrate, SN-29244, Sorafenib, Sugammadex sodium, Sunitinib malate; Tadalafil, Tafenoquine, Talnetant, Tanezumab, Tapentadol hydrochloride, Tasocitinib citrate, Technosphere/Insulin, Telcagepant, Tenofovir disoproxil fumarate, Teriparatide, Ticagrelor, Tigecycline, Tiotropium bromide, Tipifarnib, Tocilizumab, TS-041; Ulipristal acetate, Urtoxazumab, Ustekinumab; Vandetanib, Varenicline tartrate, Vicriviroc, Voriconazole, Vorinostat, VRC-HIVADV014-00-VP, VRC-HIVDNA016-00-VP; Zoledronic acid monohydrate.
3.Gateways to clinical trials.
Tomillero A1, Moral MA. Methods Find Exp Clin Pharmacol. 2010 Dec;32(10):749-73. doi: 10.1358/mf.2010.32.10.1573763.
Gateways to Clinical Trials is a guide to the most recent clinical trials in current literature and congresses. The data in the following tables has been retrieved from the Clinical Trials Knowledge Area of Thomson Reuters Integrity(SM), the drug discovery and development portal, http://www.thomsonreutersintegrity.com. This issue focuses on the following selection of drugs: 17-Hydroxyprogesterone caproate; Abacavir sulfate/lamivudine, Aclidinium bromide, Adalimumab, Adefovir, Alemtuzumab, Alkaline phosphatase, Amlodipine, Apilimod mesylate, Aripiprazole, Axitinib, Azacitidine; Belotecan hydrochloride, Berberine iodide, Bevacizumab, Bortezomib, Bosentan, Bryostatin 1; Calcipotriol/hydrocortisone, Carglumic acid, Certolizumab pegol, Cetuximab, Cinacalcet hydrochloride, Cixutumumab, Coumarin, Custirsen sodium; Darbepoetin alfa, Darifenacin hydrobromide, Darunavir, Dasatinib, Denibulin hydrochloride, Denosumab, Diacetylmorphine, Dulanermin, Duloxetine hydrochloride; Ecogramostim, Enfuvirtide, Entecavir, Enzastaurin hydrochloride, Eplerenone, Escitalopram oxalate, Esomeprazole sodium, Etravirine, Everolimus, Ezetimibe; Fenofibrate/pravastatin sodium, Ferric carboxymaltose, Flavangenol, Fondaparinux sodium; Glutamine, GSK-1024850A; Hepatitis B hyperimmunoglobulin, Hib-MenC, HIV-LIPO-5; Immunoglobulin intravenous (human), Indacaterol maleate, Indibulin, Indium 111 (¹¹¹In) ibritumomab tiuxetan, Influenza A (H1N1) 2009 Monovalent vaccine, Inhalable human insulin, Insulin glulisine; Lapatinib ditosylate, Leucovorin/UFT; Maraviroc, Mecasermin, MMR-V, Morphine hydrochloride, Morphine sulfate/naltrexone hydrochloride, Mycophenolic acid sodium salt; Naproxen/esomeprazole magnesium, Natalizumab; Oncolytic HSV; Paliperidone, PAN-811, Paroxetine, Pegfilgrastim, Peginterferon alfa-2a, Peginterferon alfa-2b/ribavirin, Pegvisomant, Pemetrexed disodium, Pimecrolimus, Posaconazole, Pregabalin; Raltegravir potassium, Ranelic acid distrontium salt, Rasburicase, Rilpivirine hydrochloride; Sertindole, Sivelestat sodium hydrate, Sorafenib, Sumatriptan succinate/naproxen sodium, Sunitinib malate; Tafluprost, Telithromycin, Temsirolimus, Tenofovir disoproxil fumavate, Tenofovir disoproxil fumarate/emtricitabine, Teriparatide, Ticagrelor, Tigecycline, Tipranavir, Tirapazamine, Trimetrexate; Ulipristal acetate; Valganciclovir hydrochloride, Vicriviroc, Vorinostat; Yttrium 90 (90Y) ibritumomab tiuxetan.
- 相关产品
-
< >
- 推荐产品
-
< >
- 最新产品
-
< >
新闻
怎么做细胞爬片免疫组化染色实验
细胞爬片免疫组化染色,是通过细胞爬片是让玻片浸在细胞培养基内,细胞在玻片上生长,主要用于组织学,免疫组织化学...
2020/7/20 22:04:33
提取病毒RNA的实验方法
提取病毒RNA方法分别有:异硫氰酸胍的提取病毒RNA方法、TRIzol LS提取法、Trizol法提取法等等...
2020/7/22 20:29:26

chelex 100树脂国产替代之路-BIOFOUNT范德生物
Chelex 100螯合离子交换树脂对铜、铁和其他重金属?的偏好显著高于对钠、钾等一价阳离子的偏好。它对二价...
2025/11/4 14:22:46

9月开学季——助研新学期 范德送好礼
2025/8/28 15:30:55

Waxfilm 实验室封口膜:技术与国际市场的双重突破
在实验室耗材领域,封口膜是保障实验准确性与稳定性的关键产品之一。近年来,Waxfilm?实验室封口膜凭借其卓...
2025/5/13 13:03:40

Waxfilm实验室封口膜的5大突破
Waxfilm实验室封口膜作为生物功能膜领域的国产技术突破和品牌突破,是生物领域中国技术发展的缩影。
2025/5/6 17:02:07

各种微流控芯片键合方法的优缺点
微流控芯片键合:目前主要有激光焊接、热压键合、胶键合、超音波焊接,每种方法都有各自的优缺点。本文主要介绍聚酯...
2023/7/28 10:43:09

新一代微流控键合解决方案
微流控键合解决方案:微流控芯片制造的一个重要环节,也是最容易被忽视的--芯片键合。其中一个重要因素是:微流控...
2023/7/27 12:44:28
荧光素钾盐使用说明
D-荧光素钾盐(K+)设计用于体外和体内生物发光测定。D-荧光素的质量和纯度对于获得良好和可重复的结果至关重...
2023/7/20 11:05:11
如何选BSA(牛血清白蛋白)
如何选BSA(牛血清白蛋白):牛血清白蛋白(BSA)有多种形式,如何选择适合自己的牛血清白蛋白(BSA)是一...
2023/2/14 13:09:18




 购物车
购物车 



