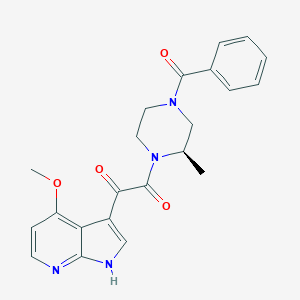
-
BMS 806
- names:
BMS 806
- CAS号:
357263-13-9
MDL Number: MFCD09026945 - MF(分子式): C22H22N4O4 MW(分子量): 406.43
- EINECS:No data available Reaxys Number:No data available
- Pubchem ID:5495818 Brand:BIOFOUNT
| 货品编码 | 规格 | 纯度 | 价格 (¥) | 现价(¥) | 特价(¥) | 库存描述 | 数量 | 总计 (¥) |
|---|
| 中文别名 | BMS 806(357263-13-9);BMS-378806;4-苯甲酰基-1-(((4-甲氧基-1H-吡咯并(2,3-b)吡啶-3-基)氧代乙酰基)-2-(R)-甲基哌嗪; |
| 英文别名 | BMS 806(357263-13-9);BMS-378806;BMS 378806;BMS378806;BMS-806;BMS806; Bms 806;4-benzoyl-1-((4-methoxy-1H- pyrrolo(2,3-b)pyridin-3-yl)oxoacetyl)-2- (R)-methylpiperazine; |
| CAS号 | 357263-13-9 |
| Inchi | InChI = 1S / C22H22N4O4 / c1-14-13-25(21(28)15-6-4-3-5-7-15)10-11-26(14)22(29)19(27)16- 12-24-20-18(16)17(30-2)8-9-23-20 / h3-9,12,14H,10-11,13H2,1-2H3,(H,23,24)/ t14- / m1 / s1 |
| InchiKey | OKGPFTLYBPQBIX-CQSZACIVSA-N |
| 分子式 Formula | C22H22N4O4 |
| 分子量 Molecular Weight | 406.43 |
| 溶解度Solubility | 生物体外In Vitro:DMSO溶解度≥ 50 mg/mL(123.02 mM)*"≥" means soluble可溶, but saturation unknown溶解度未知. |
| 性状 | Solid powder |
| 储藏条件 Storage conditions | storage at -4℃ (1-2weeks), longer storage period at -20℃ (1-2years) |
BMS 806(357263-13-9,BMS 378806)生物活性:
BMS-806(BMS 378806)是小分子gp120 / CD4抑制剂,BMS 378806具有抗感染的作用,IC50为中值5 nM。
BMS 806(357263-13-9,BMS 378806)实验注意事项:
1.实验前需戴好防护眼镜,穿戴防护服和口罩,佩戴手套,避免与皮肤接触。
2.实验过程中如遇到有毒或者刺激性物质及有害物质产生,必要时实验操作需要手套箱内完成以免对实验人员造成伤害
3.实验后产生的废弃物需分类存储,并交于专业生物废气物处理公司处理,以免造成环境污染。
Experimental considerations:
1. Wear protective glasses, protective clothing and masks, gloves, and avoid contact with the skin during the experiment.
4140. The waste generated after the experiment needs to be stored separately, and handed over to a professional biological waste gas treatment company to avoid environmental pollution.
Tags:BMS 806试剂,BMS 806杂质,BMS 806中间体,BMS 806密度,BMS 806旋光度,BMS 806闪点,BMS 806结构式,BMS 806合成,BMS 806购买,
| 产品说明 | BMS 806(357263-13-9,BMS 378806)选择性抑制HIV-1 gp120与CD4受体的结合,在病毒中的EC50为0.85-26.5 nM。 |
| Introduction | BMS 806(357263-13-9,BMS 378806)can be used as a reference substance for drug impurities and reagents,only for research. |
| Application1 | BMS 806(357263-13-9,BMS 378806)通过结合HIV包膜蛋白gp120防止HIV进入细胞。 |
| Application2 | BMS-378806是一种有效的HIV-1抑制剂,干扰CD4-gp120相互作用。BMS-378806选择性抑制HIV-1 gp120结合到CD4受体,EC50 为0.85-26.5 nM。 |
| Application3 |
BMS 806(357263-13-9,BMS 378806)药理学:
※BMS 806通过结合HIV包膜蛋白gp120防止HIV进入细胞。
※BMS 806(357263-13-9,BMS 378806)是小分子gp120 / CD4抑制剂,IC50为中值5 nM。; IC50值:5 nM;目标:HIVBMS-806(BMS 378806)被证明对HIV-1具有特异性,对HIV-2或猿猴免疫缺陷病毒没有活性。BMS-806(BMS 378806)对HIV-1分离物具有活性,而与趋化因子受体的喜好无关。BMS-806(BMS 378806)与gp120结合,特定gp120氨基酸残基的变化会改变病毒对BMS-806(BMS 378806)的敏感性。无论使用哪种特定的趋化因子受体,BMS-806(BMS 378806)都能有效抑制HIV-1感染。
| 警示图 | |
| 危险性 | warning |
| 危险性警示 | Not available |
| 安全声明 | H303吞入可能有害+H313皮肤接触可能有害+H333吸入可能对身体有害 |
| 安全防护 | P264处理后彻底清洗+P280戴防护手套/穿防护服/戴防护眼罩/戴防护面具+P305如果进入眼睛+P351用水小心冲洗几分钟+P338取出隐形眼镜(如果有)并且易于操作,继续冲洗+P337如果眼睛刺激持续+P313获得医疗建议/护理 |
| 备注 | BMS 806实验过程中防止吸入、食入,做好安全防护 |
| Wang T, et al. Discovery of 4-benzoyl-1-[(4-methoxy-1H- pyrrolo[2,3-b]pyridin-3-yl)oxoacetyl]-2- (R)-methylpiperazine (BMS-378806): a novel HIV-1 attachment inhibitor that interferes with CD4-gp120 in |
| Ho HT, et al. Envelope conformational changes induced by human immunodeficiency virus type 1 attachment inhibitors prevent CD4 binding and downstream entry events. J Virol. 2006 Apr;80(8):4017-25. |
| Guo Q, et al. Biochemical and genetic characterizations of a novel human immunodeficiency virus type 1 inhibitor that blocks gp120-CD4 interactions. J Virol. 2003 Oct;77(19):10528-36. |
| 26985308 2016-03-10 Aloperine and Its Derivatives as a New Class of HIV-1 Entry Inhibitors ACS medicinal chemistry letters |
| 23200254 2013-01-01 Inhibitors of HIV-1 attachment. Part 10. The discovery and structure-activity relationships of 4-azaindole cores Bioorganic & medicinal chemistry letters |
BMS 806(357263-13-9,BMS 378806)参考文献:
1.发现由BMS-378806驱动的靶向HIV-1 gp120-CD4相互作用的小分子抑制剂。
Liu T1, Huang B1, Zhan P2, De Clercq E3, Liu X4. Eur J Med Chem. 2014 Oct 30;86:481-90. doi: 10.1016/j.ejmech.2014.09.012. Epub 2014 Sep 6.
The HIV-1 entry into host cells is a complex, multi-factors involved, and multi-step process. Especially, the attachment of HIV-1 envelope glycoprotein gp120 to the host cell receptor CD4 is the first key step during entry process, representing a promising antiviral therapeutic target. Among the HIV-1 attachment inhibitors blocking the interaction between gp120 and CD4 cells, BMS-378806 and NBD-556 are two representative small molecular chemical entities. Particularly, BMS-378806 and its derivatives are newly identified class of orally bioavailable HIV-1 inhibitors that interfere gp120-CD4 interaction. In this review, we focused on describing the structure-activity relationships (SARs), structural modifications, in vitro or even in vivo pharmacodynamics and pharmacokinetics of BMS-378806 and its analogues as HIV-1 gp120 attachment inhibitors. In addition, the brief SARs, structural modifications of NBD-556 and its derivatives targeting the "Phe-43 cavity" as CD4 mimics were also described.
2、Molecular modeling study of HIV-1 gp120 attachment inhibitors 药物化学研究 2011年
Changdev G. Gadhe, Gugan Kothandan, Thirumurthy Madhavan & Seung Joo Cho
Abstract:The viral glycoprotein 120 (gp120) is a glycoprotein exposed on viral surface. The gp120 is essential for virus entry into cells as it plays a vital role in seeking out specific cell surface receptors for entry. In this article, we performed docking and three-dimensional quantitative structure activity relationship (3D-QSAR) study on a series of 48 indole glyoxamide derivatives as gp120 inhibitors. Docking study revealed that the inhibitor docked deeply into the gp120 cavity rather than Phe43 of cluster of differentiation 4 (CD4). 3D-QSAR methodologies, comparative molecular field analysis (CoMFA), and comparative molecular similarity indices analysis (CoMSIA) were utilized to rationalize the structural variations with their inhibitory activities. The docked pose of the most potent molecule (43) was used to determine the structures of other molecules. The CoMFA yielded a model with cross-validated correlation coefficient of (q 2) 0.73 and non-cross-validated correlation coefficient of (r 2) 0.89 with optimum number of components (N = 3). The CoMSIA models were obtained with the combination of various parameters. Final model was computed with steric, hydrophobic- and hydrogen-bond acceptor (SHA) parameters with reasonable statistics (q 2 = 0.80, r 2 = 0.94 and N = 5). The predictive power of developed CoMFA and CoMSIA models were assessed by test set (nine molecules). The predictive r 2pred for CoMFA and CoMSIA model was found to be 0.93 and 0.74, respectively. The generated contour maps were plotted onto the gp120 active site to correlate structural variations with their biological activity in protein environment. Contour map analyses showed the importance of 4-F substitution of indole ring, which made essential electronic interaction with the crucial residue (Trp427). The 3D models could explain nicely the structure–activity relationships of indole glyoxamide analogs. This would give proper guidelines to further enhance the activity of novel inhibitors.
3、Inhibitors that target gp120-CD4 interactions HIV治疗中的进入抑制剂 2007年 Pin-fang LinJohn KadowLouis Alexander
Abstract:The treatment of HIV-1 is moving towards chronic management of the disease, e.g., by combining three-drug regimens to reduce the number of dosing units. However, four classes (nucleoside/nucleotide and non-nucleoside reverse transcriptase, protease, and fusion inhibitors) including 24 approved drugs are still inadequate and treatment failures continue to occur. Factors contributing to such failures include: the emergence of drug-resistant strains, suboptimal exposure, and poor adherence that is mainly attributable to side effects. Moreover, the transmission of drug-resistant viruses is expected to rise over time. For these reasons, there is a pressing need for new classes of antiretroviral agents that are effective against HIV-1 resistant or insensitive to current drugs and that have the potential for co-formulation in convenient dosing regimens.
4、Use of the quartz crystal microbalance to monitor ligand-induced conformational rearrangements in HIV-1 envelope protein gp120 分析和生物分析化学 2009年 20016882
Hyun-Su Lee, Mark Contarino, M. Umashankara, Arne Schön, Ernesto Freire, Amos B. Smith III, Irwin M. Chaiken & Lynn S. Penn
Abstract:We evaluated the potential of a quartz crystal microbalance with dissipation monitoring (QCM-D) to provide a sensitive, label-free method for detecting the conformational rearrangement of glycoprotein gp120 upon binding to different ligands. This glycoprotein is normally found on the envelope of the HIV-1 virus and is involved in viral entry into host cells. It was immobilized on the surface of the sensing element of the QCM-D and was exposed to individual solutions of several different small-molecule inhibitors as well as to a solution of a soluble form of the host cell receptor to which gp120 binds. Instrument responses to ligand-triggered changes were in qualitative agreement with conformational changes as suggested by other biophysical methods.
5、The investigations on HIV-1 gp120 bound with BMS-488043 by using docking and molecular dynamics simulations 分子建模杂志 2012年 23086459
Liang Li, Hang Chen, Run-Ning Zhao & Ju-Guang Han
Abstract:BMS-488043, like its predecessor BMS-378806, is a small molecule that can block the interactions between gp120 and CD4, and has shown good clinical efficacy. However, the crystal structure of drug-gp120 complexes or the full-length gp120 free of bound ligand is unpublished until now. Docking combined with molecular dynamics simulation is used to investigate the binding mode between BMS-488043 and gp120. On the basis of the analysis of the simulated results, the plausible binding mode is acquired, such as the changes of binding mode in the trajectory and the calculated binding free energy. Subsequently, a number of residues which make contacts with the small molecule are studied by binding free energy decomposition to understand the mutation experiments, such as Trp427, Ser375, and Thr257 residues with the help of the acquired binding mode above. Especially, the importance of the hydrophobic groove formed by residues Ile371 and Gly472 which bind BMS-488043 is elaborated, which has not been explored much. In addition, theoretical investigations on the dynamics behavior of the gp120 associated with BMS-488043 enhanced binding are performed; the results indicate that the BMS-488043 may be more deeply inserted into the Phe43 cavity compared with the previous binding mode acquired by docking.
6、Variation in HIV-1 R5 macrophage-tropism correlates with sensitivity to reagents that block envelope: CD4 interactions but not with sensitivity to other entry inhibitors 逆转录病毒学 2008年 18205925
Paul J Peters, Maria J Duenas-Decamp, W Matthew Sullivan, Richard Brown, Chiambah Ankghuambom, Katherine Luzuriaga, James Robinson, Dennis R Burton, Jeanne Bell, Peter Simmonds, Jonathan Ball & Paul R Clapham
Abstract:HIV-1 R5 viruses cause most of the AIDS cases worldwide and are preferentially transmitted compared to CXCR4-using viruses. Furthermore, R5 viruses vary extensively in capacity to infect macrophages and highly macrophage-tropic variants are frequently identified in the brains of patients with dementia. Here, we investigated the sensitivity of R5 envelopes to a range of inhibitors and antibodies that block HIV entry. We studied a large panel of R5 envelopes, derived by PCR amplification without culture from brain, lymph node, blood and semen. These R5 envelopes conferred a wide range of macrophage tropism and included highly macrophage-tropic variants from brain and non-macrophage-tropic variants from lymph node. Results R5 macrophage-tropism correlated with sensitivity to inhibition by reagents that inhibited gp120:CD4 interactions. Thus, increasing macrophage-tropism was associated with increased sensitivity to soluble CD4 and to IgG-CD4 (PRO 542), but with increased resistance to the anti-CD4 monoclonal antibody (mab), Q4120. These observations were highly significant and are consistent with an increased affinity of envelope for CD4 for macrophage-tropic envelopes. No overall correlations were noted between R5 macrophage-tropism and sensitivity to CCR5 antagonists or to gp41 specific reagents. Intriguingly, there was a relationship between increasing macrophage-tropism and increased sensitivity to the CD4 binding site mab, b12, but decreased sensitivity to 2G12, a mab that binds a glycan complex on gp120. Conclusion Variation in R5 macrophage-tropism is caused by envelope variation that predominantly influences sensitivity to reagents that block gp120:CD4 interactions. Such variation has important implications for therapy using viral entry inhibitors and for the design of envelope antigens for vaccines. Introduction HIV-1 infection is triggered by interactions between the viral envelope glycoprotein and cell surface receptor CD4 and either of the coreceptors; CCR5 or CXCR4. These interactions induce the fusion of viral and cellular membranes and viral entry into cells. CCR5-using (R5) viruses are mainly transmitted [1], while CXCR4-using (X4) variants can be isolated from up to 50% of AIDS patients in subtype B infections and correlate with a more rapid loss of CD4+ T-cells and faster disease progression [2–5]. Among T-cells, CCR5 expression is mainly restricted to memory T-cells [6, 7], while CXCR4 is more widely expressed on various CD4+ T-cell populations including naïve T-cells [6]. R5 viruses therefore target CCR5+ memory T-cell populations and in the acute phase of replication, decimate the populations of CD4+ memory cells in lymphoid tissue associated with the gut and other mucosa [8–10]. CCR5 is also expressed on macrophage lineage cells [7] in non-lymphoid tissues e.g. the brain [11], and R5 viruses predominantly target these cells in neural tissues [12–14]. When CXCR4-using viruses emerge in late disease, they colonize naïve T-cell populations that were not infected by R5 viruses [15, 16]. Nonetheless, CD4 depletion and AIDS occur in patients from which only CCR5-using viruses can be isolated [17, 18]. In clade C infections, CXCR4-using variants have been detected in far fewer individuals in the late stages of disease [17, 19–22]. Thus, AIDS and death presumably occurs in the absence of CXCR4-using variants for a substantial number of HIV+ patients and is caused directly by R5 viruses. R5 viruses are frequently regarded as macrophage-tropic. However, several groups have reported considerable variation in the cell tropism of R5 viruses [23–25]. We reported that primary HIV-1 R5 isolates varied in their capacity to infect primary macrophage cultures by over 1000-fold [25] and we first described a subset of HIV-1 R5 isolates that could infect CD4+ T-cell lines via trace amounts of CCR5 [23]. More recently, we described R5 envelopes amplified from brain and lymph node tissue of AIDS patients that also differed markedly in tropism properties [26, 27]. Thus R5 envelopes from brain tissue were highly macrophage-tropic and were able to exploit low amounts of CD4 and/or CCR5 for infection. They contrasted considerably with R5 envelopes from immune tissue (lymph node) that conferred inefficient macrophage infection and required high amounts of CD4 for infection. Moreover, these non-macrophage-tropic envelopes were more prevalent (than macrophage-tropic envelopes) amplified from immune tissue, blood or semen [27]. These results generally support earlier reports that described a small number of highly macrophage-tropic R5 virus isolates made from brain tissue [28]. Others have confirmed that envelopes amplified from brain tissue can infect cells via low CD4 levels [29, 30]. However, Thomas et al. reported less compartmentalized variation of R5 macrophage tropism, with macrophage-tropic R5 envelopes present in both lymphoid and brain tissue [30]. The capacity of highly macrophage-tropic envelopes to use low amounts of CD4 and/or CCR5 suggests that such variants could also confer a broader tropism among CD4+ T-cells (that express low amounts of these receptors) and contribute to CD4+ T-cell depletion late in disease if they are present in immune tissue. Several groups have also reported differences in the properties of R5 virus isolates made from blood. Thus, virus isolates from late disease were reported to be more macrophage-tropic than those from earlier stages [31–33]. In addition, Repits et al. described late disease isolates with increased replicative capacity and reduced sensitivity to entry inhibitors including TAK779 (CCR5 antagonist) and T20 (gp41 inhibitor) [34]. However, they did not test whether these late isolates could exploit low CD4 or infect macrophages. It is unclear whether the highly macrophage-tropic envelopes that we have amplified from brain tissue and other sites, correspond to the late isolates described by other groups [31–34]. Recently, Dunfee et al. described a polymorphism in the C2 region of the CD4 binding site on gp120. Thus, 41% of their envelope sequences from brain tissue of patients with dementia carried an asparagine at residue 283 compared with 8% of envelopes from patients without dementia [35]. We also reported a predominance of N283 in highly macrophage-tropic brain envelopes compared to lymph node, blood and semen [27]. N283 was shown to increase the affinity of monomeric gp120 for CD4 [35]. More recently, the loss of a glycosylation site (N386) close to the CD4 binding loop on gp120 was reported to occur more frequently in HIV in the brain and was shown to contribute to increased R5 macrophage-tropism [36], an observation that we have recently confirmed (Duenas-Decamp et al. Personal communication). How variation in R5 tropism impacts on the sensitivity of HIV-1 to neutralizing antibodies and entry inhibitors is unclear. We, and others have reported that R5 macrophage-tropism correlated with increased resistance to anti-CD4 monoclonal antibodies (mabs), consistent with an increased affinity between gp120 and CD4. However, there was no correlation with sensitivity to the CCR5 antagonist, TAK779 [26, 29]. Here, we have extensively analyzed the sensitivity of thirty-six envelopes from brain, LN, blood and semen to a range of reagents that block HIV-1 entry. All these envelopes were derived from patient material by PCR without culture and have therefore not been altered by viral isolation procedures. Reagents tested for inhibition included soluble CD4 (sCD4) and tetrameric IgG-CD4 (PRO 542), BMS-378806; a small molecule that targets a site deep in the cleft that binds CD4, mabs to CD4 and CCR5, CCR5 antagonists, T20 and human mabs that recognize conserved neutralization epitopes on gp120 and gp41. Our results strongly suggest that R5 macrophage-tropism is primarily modulated by changes in the CD4 binding site on gp120 and in its affinity for CD4. Such changes impact on sensitivity to the CD4bs mab, b12 and may be driven by the presence or absence of neutralizing antibodies in vivo that target the CD4bs or proximal sites. If highly macrophage-tropic R5 variants are preferentially transmitted, then vaccines that generate antibodies to the CD4bs may be particularly effective at preventing viral transmission. Results Macrophage-tropism of brain and lymph node envelopes Envelopes used here have been described previously [26, 27] with the addition of SQ43 380.4. They are all R5, predominantly using CCR5 as a coreceptor [26, 27]. Table 1 shows macrophage infectivity as a percentage of the titer recorded on HeLa TZM-BL cells as described previously [27]. Macrophage infectivity was highly variable. Envelopes that conferred macrophage infectivity of >0.5% of infectivity for HeLa TZM-BL cells were designated as macrophage-tropic and are shown by bold script in Table 1. This arbitrary designation allows for easy identification of these envelopes as grey symbols in subsequent figures. All but one brain envelope conferred macrophage infection. None of the env+ pseudovirions carrying lymph node envelopes conferred significant macrophage infection. Macrophage-tropic R5 envelopes were amplified less frequently from blood and semen of adults and in plasma of infants.
- 相关产品
-
< >
- 推荐产品
-
< >
- 最新产品
-
< >
新闻
怎么做细胞爬片免疫组化染色实验
细胞爬片免疫组化染色,是通过细胞爬片是让玻片浸在细胞培养基内,细胞在玻片上生长,主要用于组织学,免疫组织化学...
2020/7/20 22:04:33
提取病毒RNA的实验方法
提取病毒RNA方法分别有:异硫氰酸胍的提取病毒RNA方法、TRIzol LS提取法、Trizol法提取法等等...
2020/7/22 20:29:26

chelex 100树脂国产替代之路-BIOFOUNT范德生物
Chelex 100螯合离子交换树脂对铜、铁和其他重金属?的偏好显著高于对钠、钾等一价阳离子的偏好。它对二价...
2025/11/4 14:22:46

9月开学季——助研新学期 范德送好礼
2025/8/28 15:30:55

Waxfilm 实验室封口膜:技术与国际市场的双重突破
在实验室耗材领域,封口膜是保障实验准确性与稳定性的关键产品之一。近年来,Waxfilm?实验室封口膜凭借其卓...
2025/5/13 13:03:40

Waxfilm实验室封口膜的5大突破
Waxfilm实验室封口膜作为生物功能膜领域的国产技术突破和品牌突破,是生物领域中国技术发展的缩影。
2025/5/6 17:02:07

各种微流控芯片键合方法的优缺点
微流控芯片键合:目前主要有激光焊接、热压键合、胶键合、超音波焊接,每种方法都有各自的优缺点。本文主要介绍聚酯...
2023/7/28 10:43:09

新一代微流控键合解决方案
微流控键合解决方案:微流控芯片制造的一个重要环节,也是最容易被忽视的--芯片键合。其中一个重要因素是:微流控...
2023/7/27 12:44:28
荧光素钾盐使用说明
D-荧光素钾盐(K+)设计用于体外和体内生物发光测定。D-荧光素的质量和纯度对于获得良好和可重复的结果至关重...
2023/7/20 11:05:11
如何选BSA(牛血清白蛋白)
如何选BSA(牛血清白蛋白):牛血清白蛋白(BSA)有多种形式,如何选择适合自己的牛血清白蛋白(BSA)是一...
2023/2/14 13:09:18




 购物车
购物车 



