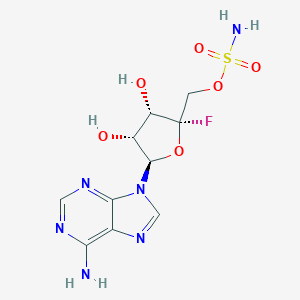
-
核杀菌素
- names:
Nucleocidin
- CAS号:
24751-69-7
MDL Number: - MF(分子式): C10H13FN6O6S MW(分子量): 364.31
- EINECS: Reaxys Number:
- Pubchem ID:72299 Brand:BIOFOUNT
| 货品编码 | 规格 | 纯度 | 价格 (¥) | 现价(¥) | 特价(¥) | 库存描述 | 数量 | 总计 (¥) |
|---|
| 中文别名 | 核杀菌素(24751-69-7);核蛋白;核苷;次氮基三乙酸-6-异硫氰酸荧光素;4'-氟-5'-O-氨磺酰腺苷;4'-C-氟代肌苷5'-氨基磺酸酯; |
| 英文别名 | Nucleocidin(24751-69-7);4'-Fluoro-5'-O-sulfamoyladenosine;4'-C-Fluoroadenosine 5'-Sulfamate;NSC 521007;nucleocidin;5'-O-Sulfamoyl-4'-fluoroadenosine;Antibiotic T-3018;Sulfamic acid 4'-fluoro-5'-adenosyl ester;T-3018;LTBCQBSAWAZBDF-XPQPFPQWSA-N;4'-Fluoro-5'-O-sulfamoyladenosine;NSC 521007; |
| CAS号 | 24751-69-7 |
| Inchi | InChI=1S/C10H13FN6O6S/c11-10(1-22-24(13,20)21)6(19)5(18)9(23-10)17-3-16-4-7(12)14-2-15-8(4)17/h2-3,5-6,9,18-19H,1H2,(H2,12,14,15)(H2,13,20,21)/t5-,6+,9-,10-/m1/s1 |
| InchiKey | LTBCQBSAWAZBDF-MLTZYSBQSA-N |
| 分子式 Formula | C10H13FN6O6S |
| 分子量 Molecular Weight | 364.31 |
| 溶解度Solubility | |
| 性状 | Not available |
| 储藏条件 Storage conditions | storage at -4℃ (1-2weeks), longer storage period at -20℃ (1-2years) |
核杀菌素(24751-69-7,Nucleocidin)毒理性质:
| 生物 | 测试类型 | 路线 | 剂量 | 影响 | 参考 |
| mouse | LD50 | oral | 2 gm/kg (2000 mg/kg) | CRC Handbook of Antibiotic Compounds, Vols.1- , Berdy, J., Boca Raton, FL, CRC Press, 1980, 5(297), 1981 | |
| mouse | LD50 | intraperitoneal | 200 ug/kg (0.2 mg/kg) | BLOOD: OTHER CHANGES | Antibiotics Annual., 4(716), 1956/1957 |
| cattle | LDLo | intramuscular | 50 ug/kg (0.05 mg/kg) | BLOOD: OTHER CHANGES | Antibiotics Annual., 4(716), 1956/1957 |
核杀菌素(24751-69-7,Nucleocidin)实验注意事项:
1.实验前需戴好防护眼镜,穿戴防护服和口罩,佩戴手套,避免与皮肤接触。
2.实验过程中如遇到有毒或者刺激性物质及有害物质产生,必要时实验操作需要手套箱内完成以免对实验人员造成伤害
3.实验后产生的废弃物需分类存储,并交于专业生物废气物处理公司处理,以免造成环境污染Experimental considerations:
1. Wear protective glasses, protective clothing and masks, gloves, and avoid contact with the skin during the experiment.
2. The waste generated after the experiment needs to be stored separately, and handed over to a professional biological waste gas treatment company to avoid environmental pollution.
| 产品说明 | 核杀菌素(24751-69-7,Nucleocidin)可以作为药物杂质对照品以及生物医药类试剂。 |
| Introduction | 核杀菌素(24751-69-7,Nucleocidin) can be used as a reference substance for drug impurities and reagents,only for research. |
| Application1 | |
| Application2 | |
| Application3 |
1、核杀菌素(24751-69-7,Nucleocidin)是一种抗生素,Nucleocidin可以抑制标签氨基酸从S-RNA转移到蛋白质的过程。
2、核杀菌素(24751-69-7,Nucleocidin)尽管在低浓度的核苷下会产生明显的抑制作用,但即使在10℃时也无法获得完全的抑制作用-3M.在可检测到核苷抑制作用之前有明显的滞后发生,并且该滞后时间的长度与核苷浓度成反比。当将亲和素从网织红细胞添加至无细胞系统时,可获得类似的结果。相比之下,在10时可获得基本完全的抑制-3嘌呤霉素仅在最低浓度时才可检测到任何滞后。核苷似乎表现出许多抗生素的典型模式,即它在氨酰基-S-RNA形成后的一个阶段抑制掺入。
| 警示图 | |
| 危险性 | warning |
| 危险性警示 | LD50 i.p. in mice: ~0.2 mg/kg (Thomas) |
| 安全声明 | H303+H313+H333 |
| 安全防护 | P264+P280+P305+P351+P338+P337+P313 |
| 备注 | 实验过程中防止吸入、食入,做好安全防护 |
| Fluorine:Chemistry,Analysis,FunctionandEffects,CHAPTER16Fluoride-InducedOxidativeStressintheLiver.SeyedFazelNabavi,MariaDaglia,AntoniSureda,SeyedMohammadNabavi,2015,Pages271-291FluorocarbonandRelatedChemistry:Volume1,AppendixII.Listofmiscellaneouspublications.R.E.Banks,M.G.Barlow,1971 |
| Volume1,Pages293-296CarbohydrateChemistry:Volume10,Nucleosides.J.S.Brimacombe,1978 |
| Volume10,Pages140-170CarbohydrateChemistry:Volume3,Introduction.R.D.Guthrie,1970 |
| Volume3,Pages3-4Fluorine:Chemistry,Analysis,FunctionandEffects,SubjectIndex,2015,Pages337-360CarbohydrateChemistry:Volume5,Halogenatedsugars.J.S.Brimacombe,1972 |
| Volume5,Pages56-60CarbohydrateChemistry:Volume3,Indexoftrivially-namedsubstances.R.D.Guthrie,1970 |
1、Nucleocidin J. R. Florini
Abstract Attention was first attracted to nucleocidin by its very great activity against trypanosomes. Subsequent investigations revealed that the toxicity of this antibiotic was too great to allow its extensive therapeutic use; consequently relatively little work has been done on its mode of action. However, its structural similarity to puromycin led to the suggestion that it might inhibit protein synthesis. Investigations of this possibility have revealed that nucleocidin is an extremely potent inhibitor of protein synthesis in vivo. In cell-free systems, however, its potency is not remarkably greater than that of other antibiotics. Although the mechanism by which nucleocidin inhibits protein synthesis has not been fully explained, some apparently unique properties of the process have been discovered.
2、Fluorinated Natural Products: Occurrence and Biosynthesis
David B. HarperEmail authorDavid O’HaganCormac D. Murphy
Abstract Despite the abundance of fluorine in the Earth’s crust naturally occurring organofluorine compounds are comparatively rare in nature with those isolated to date totalling no more than about a dozen confined to a few plants and two species of bacteria. Notwithstanding considerable interest and a variety of speculative suggestions, the mechanism of biological C-F bond formation is still unknown, although significant progress has been made in elucidating the pathway by which biosynthesis of fluoroacetate and 4-fluorothreonine occurs in the bacterium Streptomyces cattleya. In this chapter we review the nature and distribution of organofluorine compounds formed biologically and discuss the progress made in our understanding of C-F bond biosynthesis over the 60 years since fluoroacetate was first identified as a natural product.
3、Enzyme-catalyzed C–F bond formation and cleavage
Wei Tong, Qun Huang, Min Li & Jian-bo Wang
Organofluorines play an increasingly important role in the pharmaceutical and agrochemical industries, making the prospect of using enzymatic reactions to form C–F bonds bright. However, the extensive use of organofluorines has also caused environmental pollution, and thus, development of a mild green enzyme to degrade these compounds is a matter of urgency. With its highly catalytic selectivity and environmental friendliness, enzymatic catalysis will play an increasingly more important role in fluorine organic chemistry. In this review, details of the formation of C–F bonds catalyzed by fluorinase and the cleavage of C–F bonds by oxidase, reductase deaminase, and fluoroacetate dehalogenase are demonstrated. These fluorinase and defluorinase all have been isolated and identified for more than a decade, and their catalytic mechanisms illuminated. However, a narrow substrate range or low activity has hindered their application. With the fast development of biotechnology, mining new enzymes or improving their properties by directed evolution holds promise to eliminate these barriers, which will greatly accelerate the development of enzymatic organic fluorine chemistry.
4、Small-molecule inhibition of siderophore biosynthesis in Mycobacterium tuberculosis and Yersinia pestis
Julian A Ferreras, Jae-Sang Ryu, Federico Di Lello, Derek S Tan & Luis E N Quadri
Abstract Mycobacterium tuberculosis and Yersinia pestis, the causative agents of tuberculosis and plague, respectively, are pathogens with serious ongoing impact on global public health1,2 and potential use as agents of bioterrorism3. Both pathogens have iron acquisition systems based on siderophores, secreted iron-chelating compounds with extremely high Fe3+ affinity4,5. Several lines of evidence suggest that siderophores have a critical role in bacterial iron acquisition inside the human host6,7,8,9, where the free iron concentration is well below that required for bacterial growth and virulence10. Thus, siderophore biosynthesis is an attractive target in the development of new antibiotics to treat tuberculosis and plague2,5,8,11. In particular, such drugs, alone or as part of combination therapies, could provide a valuable new line of defense against intractable multiple-drug-resistant infections. Here, we report the design, synthesis and biological evaluation of a mechanism-based inhibitor of domain salicylation enzymes required for siderophore biosynthesis in M. tuberculosis and Y. pestis. This new antibiotic inhibits siderophore biosynthesis and growth of M. tuberculosis and Y. pestis under iron-limiting conditions.
5、Two 3'- O-β-glucosylated nucleoside fluorometabolites related to nucleocidin in Streptomyces calvus
Xuan Feng 1, Davide Bello 1, Phillip T Lowe 1, Joshua Clark 1, David O'Hagan
Abstract The antibiotic nucleocidin is a product of the soil bacterium Streptomyces calvus T-3018. It is among the very rare fluorine containing natural products but is distinct from the other fluorometabolites in that it is not biosynthesised from 5'-fluorodeoxyadenosine via the fluorinase. It seems to have a unique enzymatic fluorination process. We disclose here the structures of two 4'-fluoro-3'-O-β-glucosylated metabolites (F-Mets I and II) which appear and then disappear before nucleocidin production in batch cultures of S. calvus. Full genome sequencing of S. calvus T-3018 and an analysis of the putative biosynthetic gene cluster for nucleocidin identified UDP-glucose dependent glucosyl transferase (nucGT) and glucosidase (nucGS) genes within the cluster. We demonstrate that these genes express enzymes that have the capacity to attach and remove glucose from the 3'-O-position of adenosine analogues. In the case of F-Met II, deglucosylation with the NucGS glucosidase generates nucleocidin suggesting a role in its biosynthesis. Gene knockouts of nucGT abolished nucelocidin production.
- 相关产品
-
< >
- 推荐产品
-
< >
- 最新产品
-
< >
新闻
怎么做细胞爬片免疫组化染色实验
细胞爬片免疫组化染色,是通过细胞爬片是让玻片浸在细胞培养基内,细胞在玻片上生长,主要用于组织学,免疫组织化学...
2020/7/20 22:04:33
提取病毒RNA的实验方法
提取病毒RNA方法分别有:异硫氰酸胍的提取病毒RNA方法、TRIzol LS提取法、Trizol法提取法等等...
2020/7/22 20:29:26

chelex 100树脂国产替代之路-BIOFOUNT范德生物
Chelex 100螯合离子交换树脂对铜、铁和其他重金属?的偏好显著高于对钠、钾等一价阳离子的偏好。它对二价...
2025/11/4 14:22:46

9月开学季——助研新学期 范德送好礼
2025/8/28 15:30:55

Waxfilm 实验室封口膜:技术与国际市场的双重突破
在实验室耗材领域,封口膜是保障实验准确性与稳定性的关键产品之一。近年来,Waxfilm?实验室封口膜凭借其卓...
2025/5/13 13:03:40

Waxfilm实验室封口膜的5大突破
Waxfilm实验室封口膜作为生物功能膜领域的国产技术突破和品牌突破,是生物领域中国技术发展的缩影。
2025/5/6 17:02:07

各种微流控芯片键合方法的优缺点
微流控芯片键合:目前主要有激光焊接、热压键合、胶键合、超音波焊接,每种方法都有各自的优缺点。本文主要介绍聚酯...
2023/7/28 10:43:09

新一代微流控键合解决方案
微流控键合解决方案:微流控芯片制造的一个重要环节,也是最容易被忽视的--芯片键合。其中一个重要因素是:微流控...
2023/7/27 12:44:28
荧光素钾盐使用说明
D-荧光素钾盐(K+)设计用于体外和体内生物发光测定。D-荧光素的质量和纯度对于获得良好和可重复的结果至关重...
2023/7/20 11:05:11
如何选BSA(牛血清白蛋白)
如何选BSA(牛血清白蛋白):牛血清白蛋白(BSA)有多种形式,如何选择适合自己的牛血清白蛋白(BSA)是一...
2023/2/14 13:09:18




 购物车
购物车 



